We all have heard about carbohydrates even when we were in the elementary school. It is one of the basic nutrients required for proper functioning of the body. It is mainly needed for energy in the body. Most carbohydrates in the body exist in the form of polysaccharides which is also called glycan. It is called polysaccharides because it is made up of many molecules of monosaccharide that is sugars.
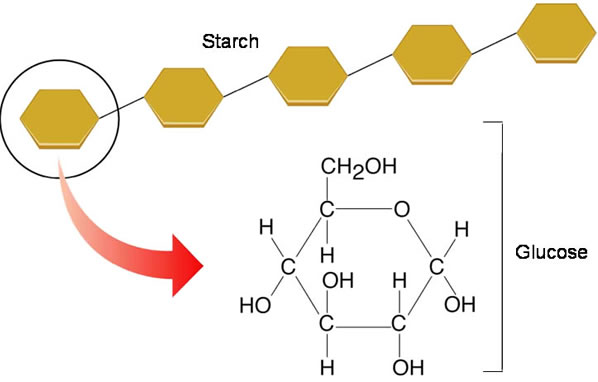
There are some polysaccharides that contain just molecules of one sugar and they called homopolysaccharides or homoglycans. Typical example of homoglycans is glycogen which is composed of glucose. Starch is another example of homoglycans but it is present in plant.
Other homoglycans are :-
- dextran which is used in present in blood plasma used in the treatment of shock
- pentosans which are made of arabinose or xylose gotten from nuts, woods and other plants
- fructans made of fructose (inulin is example of fructans)
- mannose present in ivory nuts, fungi, bacteria, pine trees and orchid tubers
- pectins that are available in berries and fruits
There are also other groups of polysaccharides that contain molecules of more than a sugar. These polysaccharides are called heteropolysaccharides or heteroglycans. Quite a good number of heteroglycans are associated with protein and they are made of two distinct units. They are called glycoproteins. Examples of such glycoproteins are acid mucopolysaccharides and gamma globulin present in the blood plasma. Heteroglycans can also be combined with lipids and these are called glycolipids. Examples of glycolipids are gangliosides available in the central nervous system.
The molecular structure of polysaccharides may be branched or linear (unbranched). Linear or branched polysaccharides can be packed together to give rise to a rigid structure. The branched structures are known for their solubility in water and they can also make pastes.
Polysaccharides can exhibit characteristics that are different from those of the monosaccharide from which they are built. While some are insoluble in water, some are soluble in water. There are even some that are amorphous.
Generally, the formula of polysaccharides is the same. It is given as Cx(H2O)y. X stands for a large number ranging from 200 to 250. The general formula of polysaccharides can also be given as (C6H10O5)n. This is because of the fact that six-carbon are the recurring units in polymer backbone.
There are some polysaccharides that are referred to as acidic polysaccharides. These are so called because they contain phosphate groups, carboxyl groups and sulphuric ester groups.
There is also another group of polysaccharides that are known as bacterial polysaccharides. These groups of glycans are available in macromolecules of different range. They include :-
- Lipopolysacharrides
- Peptidoglycan
- Exopolysaccharides
- Capsules
There are many other polysaccharides that are not listed here.
*It is good to note that polysaccharides can be used in some industrial applications.